Home
Subscription
April 2026 issue
March 2026 issue
February 2026 issue
January 2026 issue
BACK ISSUES
2025
2024
2023
2022
2021
2020
2019
2018
2017
2016
2015
2014
2013
2012
2011
2010
2009
2008
2007
2006
|
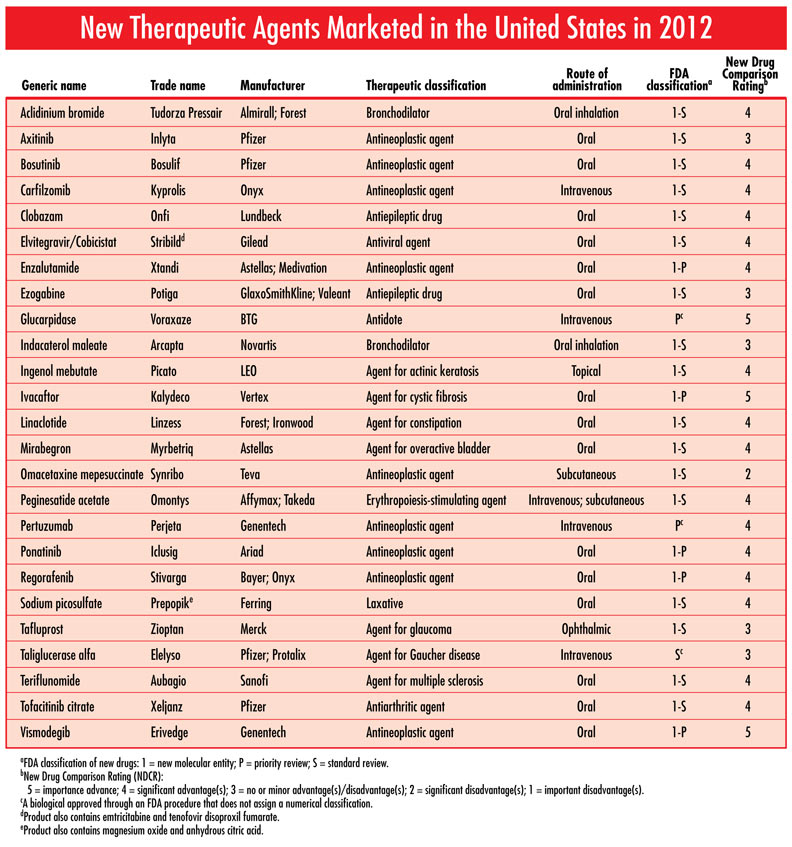
Each issue will include an editorial on a topic that is important for the profession of pharmacy, as well as a review of a new drug that includes a comparison of the new drug with previously marketed drugs that are most similar in activity, and a New Drug Comparison Rating (NDCR) for the new drug. Read on for this month's issue. January 2013 Issue [Download PDF format] In this issue: • Editorial • New Drug Review • New Therapeutic Agents Marketed in the United States in 2012 |
|||
EDITORIAL:
The APhA and ASHP Should Merge!This is the seventh editorial I have written regarding what I consider to be the extremely important need for our profession to have a much more effective organizational structure at all levels, but particularly at the national level. In the January 1996 editorial in Pharmacy Today, as well as in the January 2007, January 2009, and January 2011 editorials in The Pharmacist Activist, I voiced the opinion:"It is essential that we develop an organizational system with the size and strength to effectively address the challenges and threats to our professional responsibilities and the issue of compensation for our services...The ideal would be to have a single national pharmacy organization with the size and strength provided by a large membership base, as well as a network of divisions or academies to provide strong, effective services and representation for each pharmacy practice area." I began attending meetings of the national pharmacy associations in the late 1960s. Even then, and perhaps long before then, many pharmacists voiced strong concerns regarding the lack of "unity" within our profession. Some pharmacists urged their colleagues to place their dues to the national associations in an escrow account, and only release these funds when the associations would actively consider and make substantive progress toward the establishment of a more unified and effective national organizational structure. However, the concerns of these pharmacists were ignored by the national associations and, in the last several decades, there has been very little discussion about the organizational structure of pharmacy at the national level. Indeed, rather than moving toward a consolidation/reduction in the number of national pharmacy organizations, additional national pharmacy organizations have been established. As an alternative to addressing the organizational structure of the profession, the national associations choose to act as coalitions of associations in communicating positions on important issues, and/or participate in the Joint Commission of Pharmacy Practitioners (JCPP) that is comprised of 11 national pharmacy organizations. Quick question - How recently have you learned of a recommendation or action taken by the JCPP, and what was the issue addressed? My expectation is that most pharmacists will not have a response to this question, even though there has certainly been no lack of important issues faced by the profession of pharmacy in recent years. If those of us within the profession of pharmacy have such difficulty recalling the issues that our national associations consider important enough to discuss as a "joint commission," can we realistically expect that our legislators and others outside our profession will have a current awareness of our concerns? The successes It is not the purpose of this commentary to be critical of our national associations. Hundreds of dedicated and capable pharmacists have provided leadership and service from which our associations and profession have greatly benefited. It has been remarkable what our associations have accomplished with resources that are very limited. There have been numerous successes including pharmacist-based immunization programs, medication therapy management initiatives, and advances in patient safety, residency, fellowship, and specialty programs, to name just several. There is no question that substantial progress has been made. However, we are falling far short of our potential! The challenges Most, if not all, of our national pharmacy associations have experienced economic challenges in recent years and have had to reduce staff and implement other efficiencies. This situation is being experienced at a time when I would contend there is an unprecedented need to greatly increase financial and personnel resources to address the challenges facing pharmacy and to advance our profession. The challenges include, but are certainly not limited to, the need to greatly reduce the number of drug-related problems and errors, and greatly increase positive drug therapy outcomes for patients; integrating the role and services of pharmacists with those of physicians, physician assistants, and nurse practitioners; obtaining recognition and equitable compensation for the value of our services; contending with excessive and abusive policies and restrictions of insurance companies, PBMs, and government agencies; and recapturing our autonomy to function as health professionals. We should be encouraged by our successes but I must reluctantly conclude that we are losing our professional role and influence at a faster rate than we are making progress. However, I would hasten to say that my optimism regarding the future of our profession is sustained by the recognition that those who have the greatest need (i.e., the elderly) for the expertise and services that pharmacists are prepared to provide are the fastest growing segment of our population. Nevertheless, our profession must take bold and urgent actions to convert opportunity to reality. Strategies Several options exist for the development of a stronger national organizational structure for our profession including:
In general, I am not an advocate for "bigger is better." However, my decades of experience as an active member of our profession and multiple organizations lead me to conclude that the present structure of our national associations does not give us the strength, influence, and programs that will best serve our profession and our patients. We must make progressive change! To move in the direction of merging the national pharmacy associations, my first thought was to initially encourage the smaller national associations that had some overlapping goals and programs to actively consider merging their associations. However, while certainly not wanting to discourage such discussions, I recommend that we start this initiative with our largest associations, specifically APhA and the American Society of Health-System Pharmacists (ASHP). The APhA and ASHP have an excellent opportunity to provide leadership that will strengthen the structure and effectiveness of our profession through organizational change in a manner that would make it attractive for the other national pharmacy associations to also want to be a part of this process. As one pharmacist who has been a member of both APhA and ASHP for many years, I can think of nothing else that would give me as much encouragement and optimism for the future of our profession than if APhA and ASHP would merge and attain the greater strength and influence that are so important for our profession to advance. There are thousands of pharmacists who presently do not belong to even one national pharmacy association, and I believe that the enthusiasm surrounding the initiative to create a more effective national organizational structure would result in many of these pharmacists supporting this effort with their membership and participation. We should encourage the leaders of both APhA and ASHP to begin discussions regarding the national organizational structure for pharmacy and the potential to merge APhA and ASHP. I do not underestimate the scope and implications of this effort, and the loyalties and passions that support each of these associations. Others may have better recommendations but, at present, the alternative appears to be maintaining the status quo. That is not sufficient. Bold action is needed now! Daniel A. Hussar |
NEW DRUG REVIEW:
Aclidinium bromide
New Drug Comparison Rating (NDCR) = 4 | ||
|
|||